商品描述
McKesson # 237000
Manufacturer # 00409488710
Manufacturer Hospira
Country of Origin Unknown
Alternate Manufacturer Number 2718062
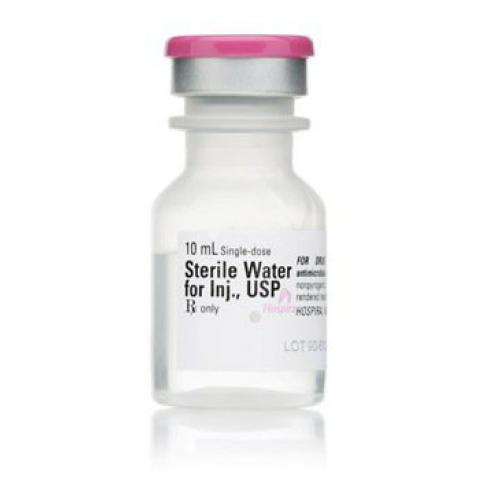
Application Diluent
Container Type Single Dose Vial
Dosage Form Injection
Generic Drug Code 02709
Generic Drug Name Sterile Water for Injection, Preservative Free
NDC Number 00409-4887-10
Product Dating McKesson Acceptable Dating: we will ship >= 90 days
Storage Requirements USP Controlled Room Temperature
UNSPSC Code 51191605
Volume 10 mL
Latex Free Indicator Not Made with Natural Rubber Latex
Features
◾ Single dose plastic flip-top vial
◾ This preparation is designed solely for parenteral use only after addition of drugs that require dilution or must be dissolved in an aqueous vehicle prior to injection
◾ It contains no bacteriostat, antimicrobial agent or added buffer and is supplied only in single-dose containers to dilute or dissolve drugs for injection